Radioactivity - the release of nuclear radiation in teh form of paricles and rays from a radioactive element.
If an element is radioactive is has an unstable nucleus; needs to go under nuclear reactions to become stable.
Nuclear reactions involve the atoms nucleus
- The nucleus is opened and the protons and neutrons are rearranged
- Founded by Marie and Pierre Curie
- Marie came up with the phrase radioactivity
- They found 2 radioactive elements ( Po, and Ra )
If an element is radioactive is has an unstable nucleus; needs to go under nuclear reactions to become stable.
Nuclear reactions involve the atoms nucleus
- The nucleus is opened and the protons and neutrons are rearranged
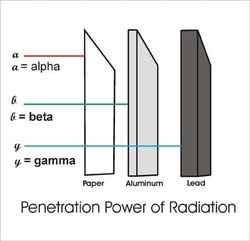
Types of Radiation
- Alpha (á) - which is a positively charged helium isotopes (Weakest). Consists of helium nuclei released from a radioactive source.
- Beta (B) - an electron (Middle) an electron result from the breaking apart of a neutron in an atom.
- Gamma (y) - pure energy; called a ray rather than a particle (Strongest). High energy photon, electromagnetic. Nuclei often release gamma rays with alpha or beta particles during radioactive decay. It has no mass or charge.
Balancing Nuclear reactions…
In the reactants (on the left) and products (on the right), the atomic and mass numbers MUST BALANCE
Use a particle or isotope to fill in missing protons and neutrons.
In the reactants (on the left) and products (on the right), the atomic and mass numbers MUST BALANCE
Use a particle or isotope to fill in missing protons and neutrons.
Information was presented in class.
