Heat vs. Temperature
Heat (q)- a form of energy, usually measured in Joules. Energy is transferred between samples of matter because of the difference in their temps.
Temperature- a measure of the AVERAGE KINETIC ENERGY of the particles in a sample of matter.
- Makes molecules move faster ( increases Temperature )
- gives molecules more freedome ( solid--> liquid--> gas)
Temperature- a measure of the AVERAGE KINETIC ENERGY of the particles in a sample of matter.
Specific Heat
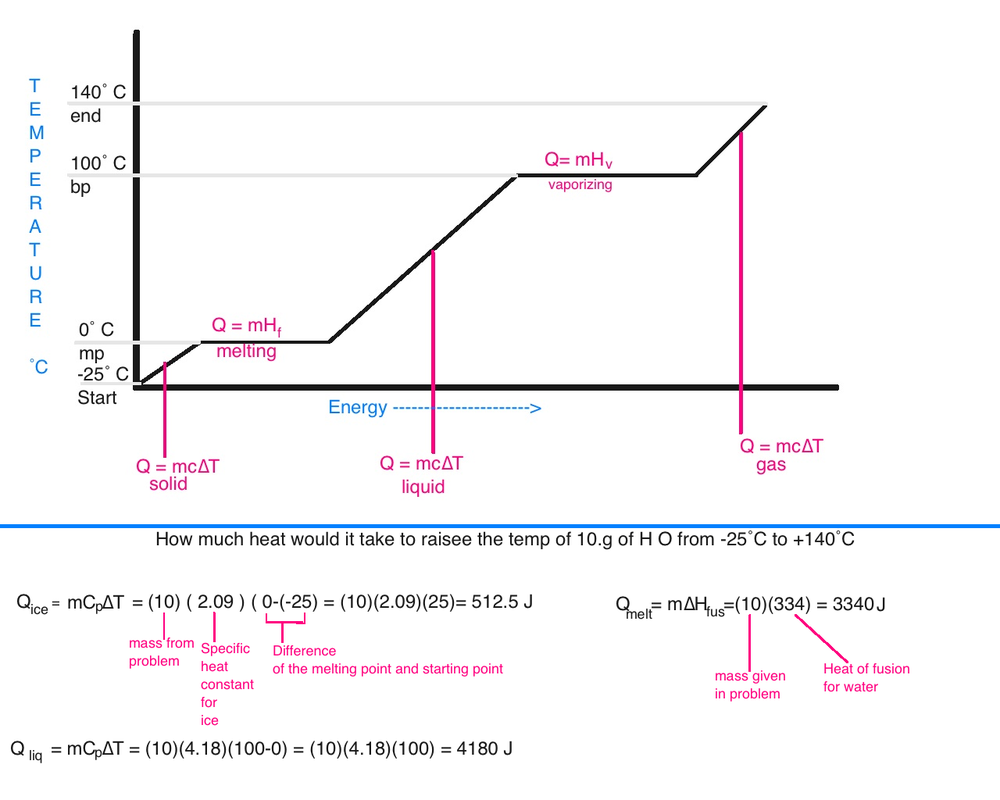
Specific Heat- the amount of heat (q) that it takes to increase the temp of 1 gram of a substance by 1 ˚C . For a given mass (m), specific heat ( Cp ) and Temperature Change (∆ T )
q = m (Cp) ( ∆ T)
specific heat for ice is 2.09 J/g ˚C
Specific heat for water is 4.184 J/g˚C
Specific heat for water vapor 1.97 J/g˚C
Heat of fusion for water is 334 J/G
Heat of vaporization for water is 2260 J/G
q = m (Cp) ( ∆ T)
specific heat for ice is 2.09 J/g ˚C
Specific heat for water is 4.184 J/g˚C
Specific heat for water vapor 1.97 J/g˚C
Heat of fusion for water is 334 J/G
Heat of vaporization for water is 2260 J/G
Specific heat CAPACITY ( shc)
Physical property of a substance used to compare heat absorption capabilities for different materials. Measured in J/gK
Specific heat ( cp ) the subscript 'p' stands for pressure
pressure = force per unit area on a surface
Specific heat ( cp ) the subscript 'p' stands for pressure
pressure = force per unit area on a surface
Endo or Exo- thermic Reaction?
Another way to stat 'q' energy gained/ lost during the reaction is ∆H, which stands for enthalpy change ( another physical property. )
∆H ( enthalpy ) is the difference between stored energy of the reactants an rxtn in the reaction.
∆H ( enthalpy ) is the difference between stored energy of the reactants an rxtn in the reaction.
- if ∆H is negative, the reaction is EXOTHERMIC
- if ∆H is positive, the reaction is ENDOTHERMIC.
- most reactions are exothermic, occurring spontaneously.
- Exothermic reactions can also occur spontaneously because of entropy.
- Entropy = the degree of randomness of particles in a system.
- +∆S in a reaction = normal, -∆S is RARE.
Information was presented in class.