Properties of Acids
- Produs H+ ( as H3O+ ) ions in water
- Taste sour
- Corrode metals
- Electrolytes
- When they react with bases they from salt and water
- pH is LESS than 7
- Turns blue litmus paper to red
Naming Acids--Hydrogen /nonmetal
- If not in water 1. hydrogen 2. name of the anion
- if it is in water ( aq ) use the prefix "hydro' - root - 'ic" acid
Naming Acids-- Hydrogen/Polyatomic anion
- not in water 1. hydrogen 2. name of anion
- in water - root name then ending change on anion…. -ite ending turns to -ous acid. -ate ending turns to - ic acid
Properties of Bases
- Produce OH- ions in water
- Taste bitter, chalky
- Are electrolytes
- Feel soapy, slippery
- React with acids to form salts and water.
- pH is greater than 7
- Turns red litmus paper blue
Acid/base Definitions
Definition #1: Arrhenius ( traditional )
Definition #2: Brønsted- Lowry
Definition #3: Lewis
- Acids- produce H+ ions
- Bases - Produce OH- ions ( not every base is in the OH- group )
Definition #2: Brønsted- Lowry
- Acids - proton donor
- Bases - Proton acceptors
Definition #3: Lewis
- Lewis acid- a substance that accepts an electron pair
- Lewis bas- a substance that donates an electron pair
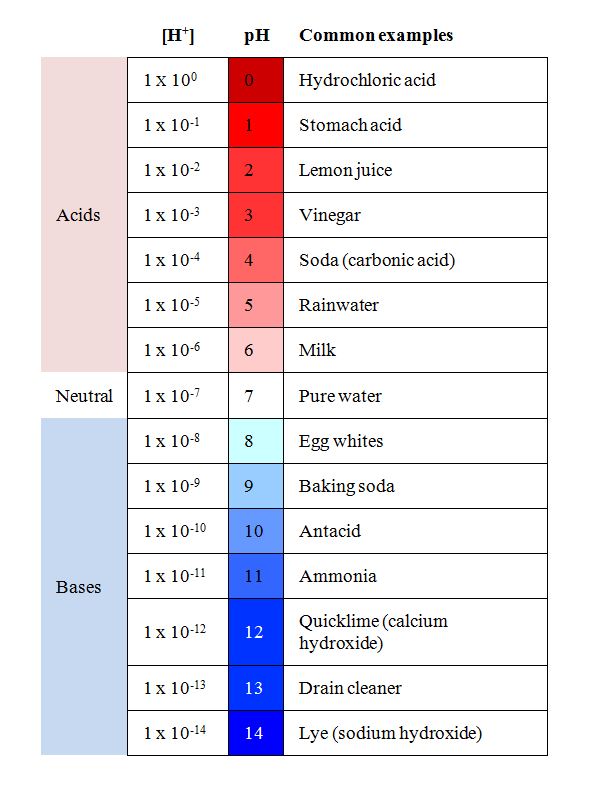
pH
- [H+] is critical in many chemical reactions
- pH= -log[H+], [H+]= 10^-pH
- pH scale is from 1-14
- substance > 7 = base
- substance < 7 = acid
- substance = 7 = neutral
Information was presented in class.